Single dose of zilebesiran safely and effectively lowered blood pressure for six months
[ad_1]
Research Highlights:
- The phase 2 study found that a single injection of the experimental medication zilebesiran safely and effectively lowered blood pressure in adults with mild-to-moderate hypertension for up to six months.
- Based on these results, zilebesiran has the potential to improve medication adherence due to infrequent dosing, which will, in turn, reduce cardiovascular risk in people with hypertension.
 Embargoed until 4 p.m. ET, Saturday, Nov. 11, 2023
Embargoed until 4 p.m. ET, Saturday, Nov. 11, 2023
(NewMediaWire) – November 11, 2023 – PHILADELPHIA — A single injection of the experimental medication zilebesiran was safe and effective in reducing systolic blood pressure in people with mild-to-moderate high blood pressure for up to six months, as detailed in the Phase 2 of the KARDIA study, reported as late-breaking science today at the American Heart Association’s Scientific Sessions 2023. The meeting, Nov. 11-13, in Philadelphia, is a premier global exchange of the latest scientific advancements, research and evidence-based clinical practice updates in cardiovascular science.
Zilebesiran is an investigational RNA interference agent targeting angiotensinogen (AGT), a hormone produced predominantly in the liver that contributes to blood pressure regulation.
“Uncontrolled high blood pressure is a leading cause of death and disease, so there is a need for new treatments that provide sustained blood pressure control over longer periods of time. This will improve outcomes for people with hypertension,” said lead study author George L. Bakris, M.D., FAHA, a professor of medicine and director of the Comprehensive Hypertension Center at the University of Chicago Medicine. “Uncontrolled hypertension also increases the risk of heart disease.”
According to the American Heart Association’s 2023 Statistical Update, heart disease continues to be the number one cause of death in the United States and nearly half of adults in the U.S. have high blood pressure.
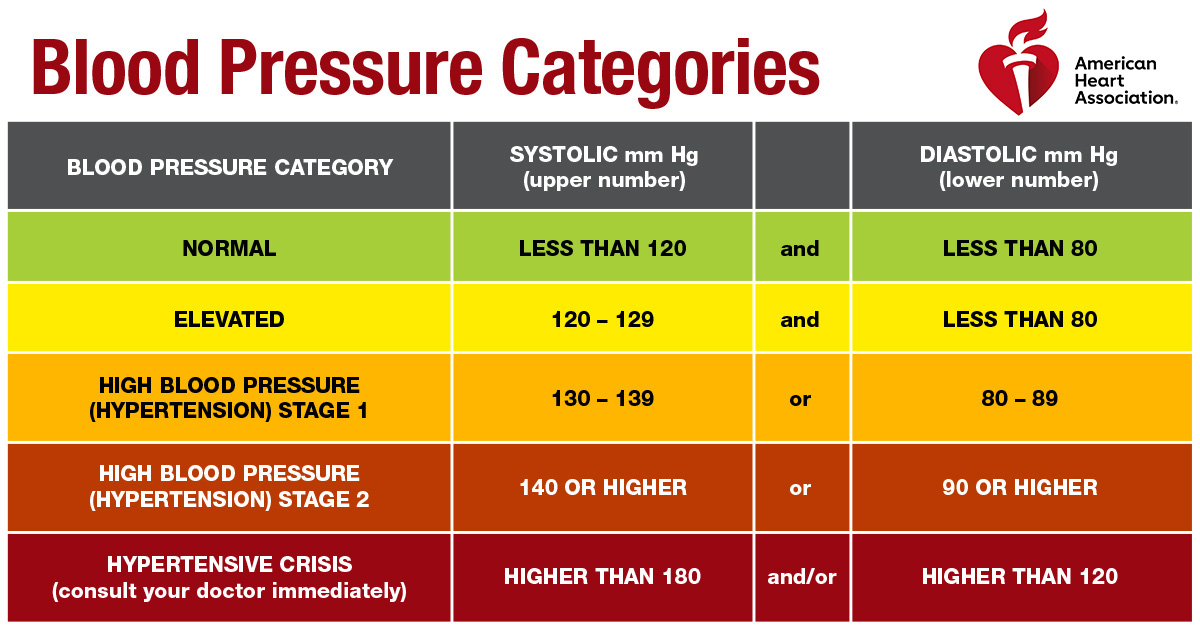
In this global, placebo-controlled, randomized, double-blind trial, researchers investigated the safety and efficacy of administering zilebesiran to people with mild-to-moderate high blood pressure, defined as systolic blood pressure of 135-160 mm Hg, who are untreated or on stable therapy with up to two antihypertensive medications. Systolic blood pressure is the top number in a blood pressure reading and indicates how much pressure blood is exerting against artery walls when the heart contracts.
The 2017 ACC/AHA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults classifies stage 1 hypertension as having top and bottom blood pressure measures greater than or equal to 130/80 mm Hg, and stage 2 hypertension as having top and bottom measures greater than or equal to 140/90 mm Hg.
The study included 394 patients with an average systolic blood pressure of 142 mm Hg. Participants were randomized to receive subcutaneous doses of zilebesiran (4 groups: 150, 300 or 600 mg once every six months or 300 mg once every three months) or a placebo.
Over the six-month study period, the analysis found:
- Compared to participants who received the placebo, participants who received single doses of zilebesiran had greater than 10 mm Hg reductions, on average, in 24-hour systolic blood pressure and greater than 90% reductions in serum levels of AGT, a hormone produced predominantly in the liver that contributes to blood pressure regulation.
- At the three-month follow-up, the participants in groups that received the 300 mg and 600 mg doses of zilebesiran had 24-hour average systolic blood pressure that was lowered by 15 mm Hg or more on average.
- After six months, people receiving zilebesiran were significantly more likely to experience 24-hour average systolic blood pressure reductions of 20 mm Hg or more on average without taking additional high blood pressure medications.
- People receiving zilebesiran were more likely to achieve 24-hour average systolic blood pressure measurements of 130 mm Hg or less at six months.
In addition, participants in all four zilebesiran groups consistently experienced significantly greater reductions in both daytime and nighttime systolic blood pressure. Low rates of zilebesiran-related adverse events were reported, with the most common being mild reactions at the site of the injection. No clinically relevant changes in kidney or liver function were observed, Bakris noted.
“Our study demonstrates that either quarterly or biannual doses of zilebesiran can effectively and safely lower blood pressure in patients with uncontrolled hypertension,” Bakris said. “It is well known that reductions in systolic blood pressure of greater than or equal to 5 mm Hg are linked to a reduction in cardiovascular risk. These results reinforce the potential of zilebesiran to provide sustained blood pressure control, improve adherence to medication via infrequent dosing, and in turn, improve outcomes for people with high blood pressure.”
Study details and background:
- 394 patients were randomized, and 377 patients were ultimately included in the analysis: 302 were randomized to one of the four zilebesiran dosage groups and 75 people to the placebo group. Their average age was 57 years old.
- About 25% of participants were Black adults; 56% were men.
- The study was conducted at sites in Canada, Ukraine, the United Kingdom and the U.S., and 78% of participants lived in the U.S. Due to challenges with data collection with the war in the Ukraine, data from the 16 patients randomized in Ukraine were excluded from the analyses.
- Average blood pressure of participants at time of enrollment in the study was 141.8/81.8 mm Hg.
- There were four non-serious, related adverse reactions leading to discontinuation in the zilebesiran groups: two instances of orthostatic hypotension; one of blood pressure elevation; and one of injection site reaction. There were no adverse reactions in the placebo group.
- The six-month, placebo-controlled treatment period of the Phase 2 trial reported here was conducted from July 2021 to June 2023.
These findings are limited because the KARDIA-1 trial evaluated zilebesiran only in people with mild-to-moderate hypertension. Another study limitation is that results are limited to the six-month placebo-controlled period. Longer-term safety and impact on cardiovascular outcomes will be assessed in future research.
Co-authors, disclosures and funding sources are listed in the abstract.
Statements and conclusions of studies that are presented at the American Heart Association’s scientific meetings are solely those of the study authors and do not necessarily reflect the Association’s policy or position. The Association makes no representation or guarantee as to their accuracy or reliability. Abstracts presented at the Association’s scientific meetings are not peer-reviewed, rather, they are curated by independent review panels and are considered based on the potential to add to the diversity of scientific issues and views discussed at the meeting. The findings are considered preliminary until published as a full manuscript in a peer-reviewed scientific journal.
The Association receives funding primarily from individuals; foundations and corporations (including pharmaceutical, device manufacturers and other companies) also make donations and fund specific Association programs and events. The Association has strict policies to prevent these relationships from influencing the science content. Revenues from pharmaceutical and biotech companies, device manufacturers and health insurance providers and the Association’s overall financial information are available here.
Additional Resources:
About the American Heart Association
The American Heart Association is a relentless force for a world of longer, healthier lives. We are dedicated to ensuring equitable health in all communities. Through collaboration with numerous organizations, and powered by millions of volunteers, we fund innovative research, advocate for the public’s health and share lifesaving resources. The Dallas-based organization has been a leading source of health information for nearly a century. Connect with us on heart.org, Facebook, X or by calling 1-800-AHA-USA1.
###
For Media Inquiries and AHA Expert Perspective:
AHA Communications & Media Relations in Dallas:
John Arnst:
For Public Inquiries: 1-800-AHA-USA1 (242-8721)
heart.org and stroke.org
[ad_2]