BIO Illustrates the Success of the Orphan Drug Act
[ad_1]
The numbers may shock you. Approximately 7,000 uncommon ailments are identified to exist, and roughly 30 new ones are recognized every year. Also generally known as orphan ailments, they’re outlined as these which have an effect on fewer than 200,000 sufferers in America. And whereas it may be true that every uncommon illness solely impacts a small quantity of sufferers, collectively, thousands and thousands of lives are relying on the improvement of revolutionary cures and medicines.
In a collection of new infographics, BIO illustrates how far we’ve come – and the way far we have now to go – in the combat towards uncommon ailments. You can view them here and here.
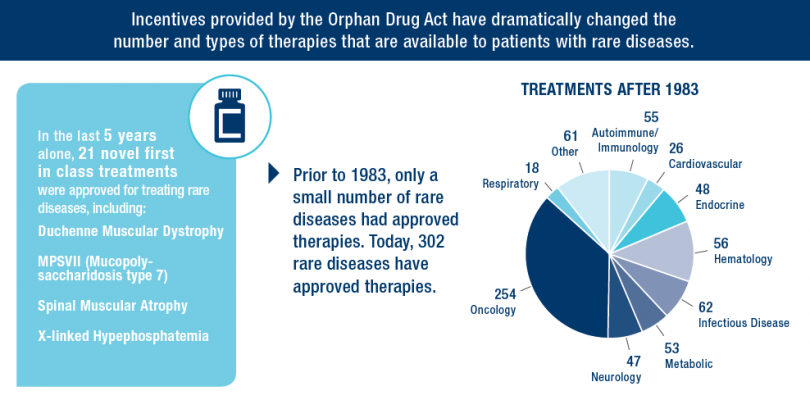
The Orphan Drug Act has given households and people affected by uncommon ailments like Hodgkin Lymphoma and Cystic Fibrosis hope and optimism. Since the legislation’s passage in 1983, tons of of new therapies have been authorized by the FDA to deal with these uncommon ailments—in comparison with simply 34 approvals earlier than enactment.
Outstanding scientists, savvy entrepreneurs and enterprise leaders, a dedicated funding neighborhood and world-class universities and analysis establishments are essential to develop breakthrough medicines and cures. But as the success of the Orphan Drug Act illustrates, the proper public coverage atmosphere and incentives are additionally important in spurring innovation for uncommon ailments. In order to construct on the historic successes of the Orphan Drug Act, is important that the incentives offered by the Act are stored in place to encourage improvement of therapies for uncommon ailments.
To be taught extra, go to www.bio.org/toolkit/disease-area.
[ad_2]